Laguna Announces FDA Clearance of IND Application for LGNA-100, a Novel γδ T Cell Activator for High
SAN FRANCISCO, April 29, 2026 (GLOBE NEWSWIRE) -- Laguna Biotherapeutics, Inc. (Laguna), a clinical-stage biotechnology company focused on novel live bacterial therapeutics, today announced that the U.S. Food and Drug Administration (FDA) has cleared its Investigational New Drug (IND) application, granting a "safe to proceed" for its lead clinical candidate from the QUAIL platform, LGNA-100.
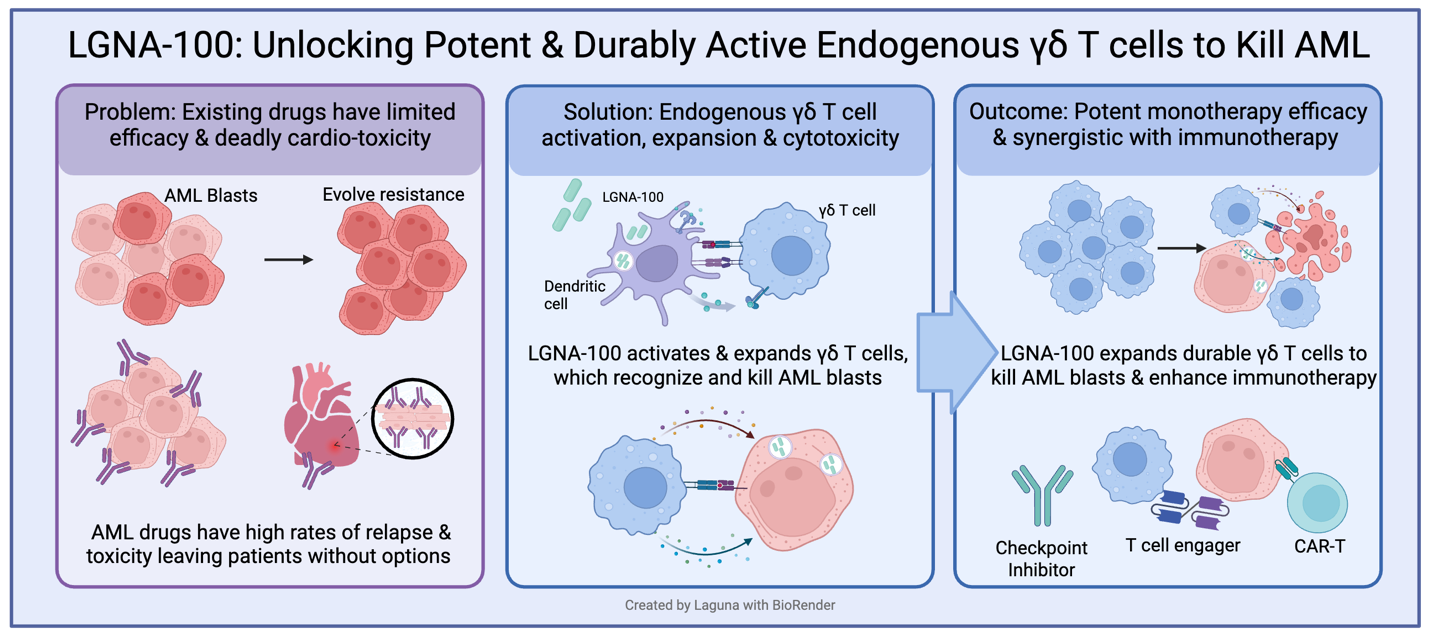
LGNA-100 is a first-in-class, attenuated live bacterial immunotherapy designed to safely harness our immune system's evolved response to Listeria; robustly and durably expanding and activating endogenous γδ T cells that can directly kill cancer cells while also improving existing immunotherapies. The Phase 1 first-in-human study will validate the QUAIL platform and evaluate LGNA-100 in patients with high-risk leukemia following hematopoietic stem cell transplantation (HSCT) to prevent leukemic relapse.
"The IND clearance of our first clinical study is a defining moment and transformative milestone for Laguna as we transition into a clinical-stage company," said Jonathan Kotula, Ph.D., CEO of Laguna. "Our goal is to create systems-level therapies for complex diseases. With LGNA-100, and the QUAIL platform we are taking a fundamentally new approach to selectively stimulate innate T cells to improve long-term outcomes for pediatric patients with high-risk leukemia."
The clinical rationale for the QUAIL platform builds directly upon decades of research into the human γδ T cell response to Listeria, and the protective role of γδ T cells against leukemia recurrence following HSCT. A presentation covering our clinical rationale will be presented at the ISCT 2026 Annual Meeting on May 6 in Dublin, Ireland.
"In the setting of αβ-depleted HSCT, γδ T cells are critical effectors that provide potent graft-versus-leukemia activity without driving graft-versus-host disease," said Dr. Alice Bertaina, MD, PhD, Co-Director of the Bass Center for Childhood Cancer and Blood Diseases, Lucile Packard Children's Hospital at Stanford University and lead Clinical Advisor for Laguna. "While early pharmacologic activators like zoledronic acid (ZOL) showed the clinical potential of this approach, intense ZOL stimulation pushes these cells into a more mature, terminally differentiated and exhausted state. Our comprehensive preclinical evaluations demonstrate that LGNA-100 drives a distinct, multifunctional γδ T cell response with improved kinetic, phenotypic, and functional features compared to ZOL, supporting more durable activation without hyperactivation or early exhaustion. I am very excited to see this translated into the clinic for these high-risk leukemia patients."
"Pediatric AML remains one of the most challenging frontiers in oncology, demanding novel modalities that can detect and kill the disease without compounding toxicities," said Bill Newell, former CEO of Sutro Biopharma and Strategic Advisor to Laguna. "Having spent years evaluating platforms to tackle these exact malignancies, I believe Laguna's approach using γδ T cells to potentially solve the problems associated with high-risk leukemia is a massive leap forward. Securing this IND is a testament to the rigor of their science and positions LGNA-100 as a highly differentiated asset in the cancer immunotherapy space."
The Phase 1 clinical study is a company-sponsored open-label, first-in-human, single ascending dose study designed to assess safety and tolerability, and support the proof of LGNA-100's mechanism of action. The study will evaluate LGNA-100 administered via intravenous (IV) infusion in pediatric and young adult participants with high-risk acute leukemias and MDS who have received an αβ-depleted HSCT.
About LGNA-100
LGNA-100 also known as QUAIL-100 is an investigational cancer immunotherapeutic agent derived from Listeria monocytogenes (Lm), developed from the QUAIL platform, designed to activate and expand a patient's endogenous γδ T cells.
About Laguna Biotherapeutics, Inc. (Laguna)
Laguna is a privately funded, clinical-stage biotechnology company headquartered in San Francisco, CA with research labs in Siena, Italy. It is focused on developing novel therapeutics that leverage Lm to activate and expand the immune system to create impactful treatments across oncology, as well as inflammatory and infectious disease.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/14da867a-1402-4d3d-b473-d3267f862b39

热门文章
L'ATELIER DE JOËL ROBUCHON HONG KONG 延续十八载辉煌传奇 今夏载誉回归置地廣塲
2025年TOURISE大奖揭晓:东京、纽约、安卡什与巴黎荣获首届殊荣
从“眼巴巴”到“手飒飒”,5年59次打破国外技术垄断——国家管网集团北京管道有限公司“红色劲旅自主维保突击队”攻坚实录
2025年度全国会计专业技术中级资格考试 (甘肃考区)报名公告
「全球能源循环计划」愿景发布 电池循环经济加速落地
品誉咨询——绩效破局:从 “效率优先” 到 “效能质变” 的管理范式革新